For adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to or intolerance of one or more TNF blockers. Use Restriction: Upadacitinib is not recommended for concomitant use with other JAK inhibitors, biological DMARDs, or potent immunosuppressants (e.g., azathioprine and cyclosporine). For adult patients with active psoriatic arthritis who have had an inadequate response to or intolerance of one or more TNF blockers. Use Restriction: Upadacitinib is not recommended for concomitant use with other JAK inhibitors, biological DMARDs, or potent immunosuppressants (e.g., azathioprine and cyclosporine). For adult and pediatric patients aged 12 years and older with refractory, moderately to severely active atopic dermatitis whose disease is not adequately controlled with other systemic medications (including biologics) or for whom such therapies are not advisable. Use Restriction: Upadacitinib is not recommended for concomitant use with other JAK inhibitors, biological immunomodulators, or other immunosuppressants. For adult patients with moderately to severely active ulcerative colitis who have had an inadequate response to or intolerance of one or more TNF blockers. Use Restriction: Upadacitinib is not recommended for concomitant use with other JAK inhibitors, biological therapies for ulcerative colitis, or potent immunosuppressants (e.g., azathioprine and cyclosporine). For adult patients with moderately to severely active Crohn's disease who have had an inadequate response to or intolerance of one or more TNF blockers. Use Restriction: Upadacitinib is not recommended for concomitant use with other JAK inhibitors, biological therapies for Crohn's disease, or potent immunosuppressants (e.g., azathioprine and cyclosporine). For adult patients with active ankylosing spondylitis who have had an inadequate response to or intolerance of one or more TNF blockers. Use Restriction: Upadacitinib is not recommended for concomitant use with other JAK inhibitors, biological DMARDs, or potent immunosuppressants (e.g., azathioprine and cyclosporine). For adult patients with active non-radiographic axial spondyloarthritis with objective signs of inflammation who have had an inadequate response to or intolerance of TNF blocker therapy. Use Restriction: Upadacitinib is not recommended for concomitant use with other JAK inhibitors, biological DMARDs, or potent immunosuppressants (e.g., azathioprine and cyclosporine).
乌帕替尼缓释片Upadacitinib 15mg


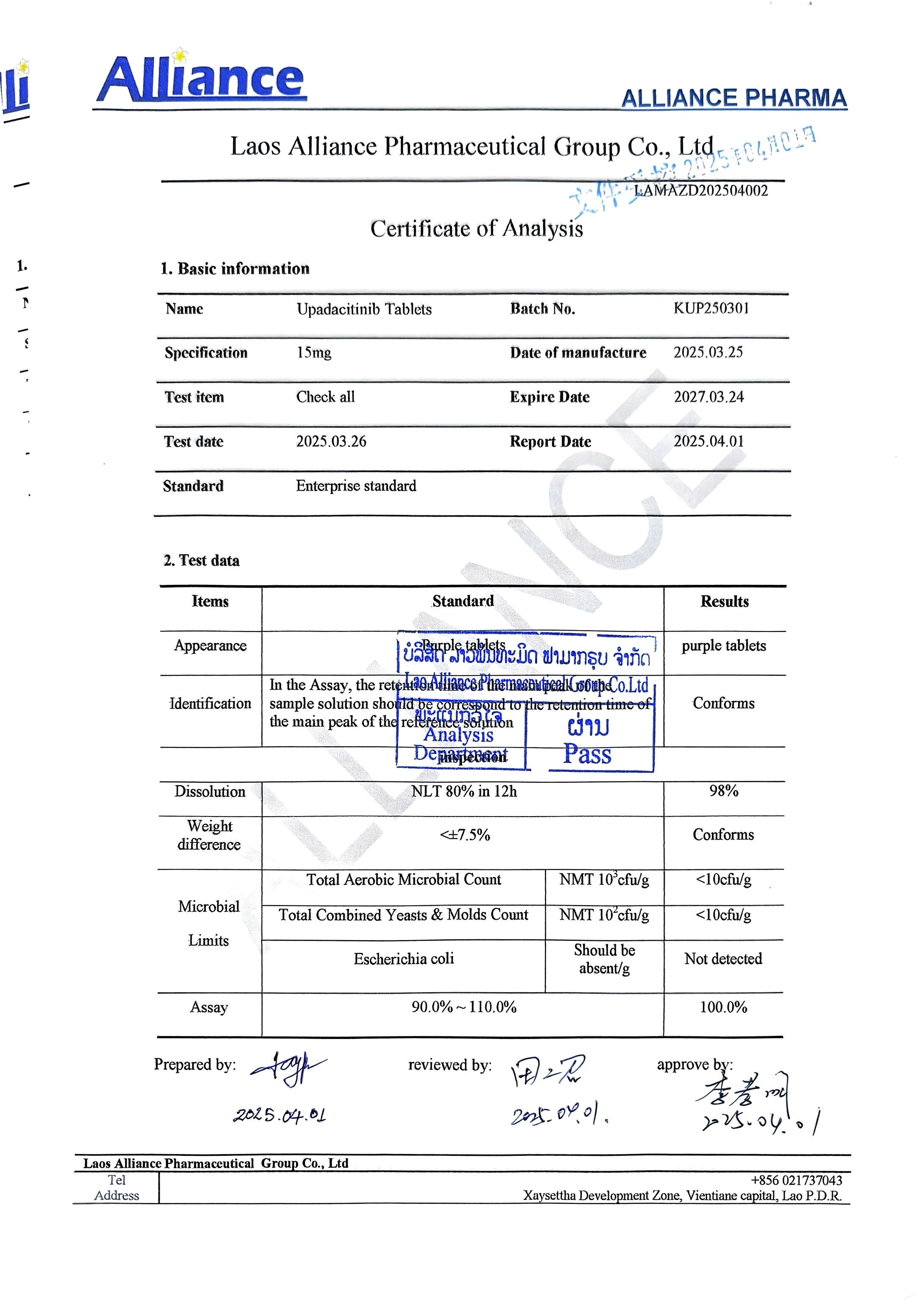


¥380.00
售价:¥380.00
销量:886 件
Rheumatoid Arthritis, Psoriatic Arthritis, Ankylosing Spondylitis, and Non-radiographic Axial Spondyloarthritis Recommended dose: 15 mg once daily. Atopic Dermatitis For pediatric patients aged 12 years and older weighing at least 40 kg and adults under 65 years of age: The recommended initial dose is 15 mg orally once daily. If an adequate response is not achieved, consider increasing the dose to 30 mg orally once daily. For adults 65 years of age and older: Recommended dose is 15 mg once daily. For patients with severe renal impairment: Recommended dose is 15 mg once daily. Ulcerative Colitis For adults: Recommended induction dose is 45 mg once daily for 8 weeks. Recommended maintenance dose is 15 mg once daily. For patients with refractory, severe, or extensive disease, a maintenance dose of 30 mg once daily may be considered. Discontinue upadacitinib if the 30 mg dose does not provide an adequate therapeutic response. Use the lowest effective dose needed to maintain response. Crohn's Disease For adults: Recommended induction dose is 45 mg once daily for 12 weeks. Recommended maintenance dose is 15 mg once daily. For patients with refractory, severe, or extensive disease, a maintenance dose of 30 mg once daily may be considered. Discontinue upadacitinib if the 30 mg dose does not provide an adequate therapeutic response. Use the lowest effective dose needed to maintain response.
Patients with hypersensitivity to the active ingredient of this product or any of its excipients. Active tuberculosis or active severe infections (including localized infections). Severe hepatic impairment. Pregnancy.
Severe Infections: Avoid use in patients with active severe infections, including localized infections. Gastrointestinal (GI) Perforation: Monitor for signs and symptoms of GI perforation. Laboratory Monitoring: It is recommended to monitor changes in lymphocytes, neutrophils, hemoglobin, liver enzymes, and lipids. Pregnancy and Lactation: Administration during pregnancy may cause fetal harm. Women who are breastfeeding should discontinue breastfeeding during treatment. Contraceptive measures should be taken during the period of medication. Vaccination: Avoid concomitant use with live vaccines.
Upper respiratory tract infections, herpes zoster, herpes simplex, bronchitis, nausea, cough, fever, acne, headache, hypersensitivity reactions, folliculitis, abdominal pain, elevated blood creatine, etc.
15 mg per tablet; 28 tablets per bottle.
Store in a sealed container, below 30℃. Keep out of the reach of children.
评论
添加评论
请登录后发表评论
暂无评论