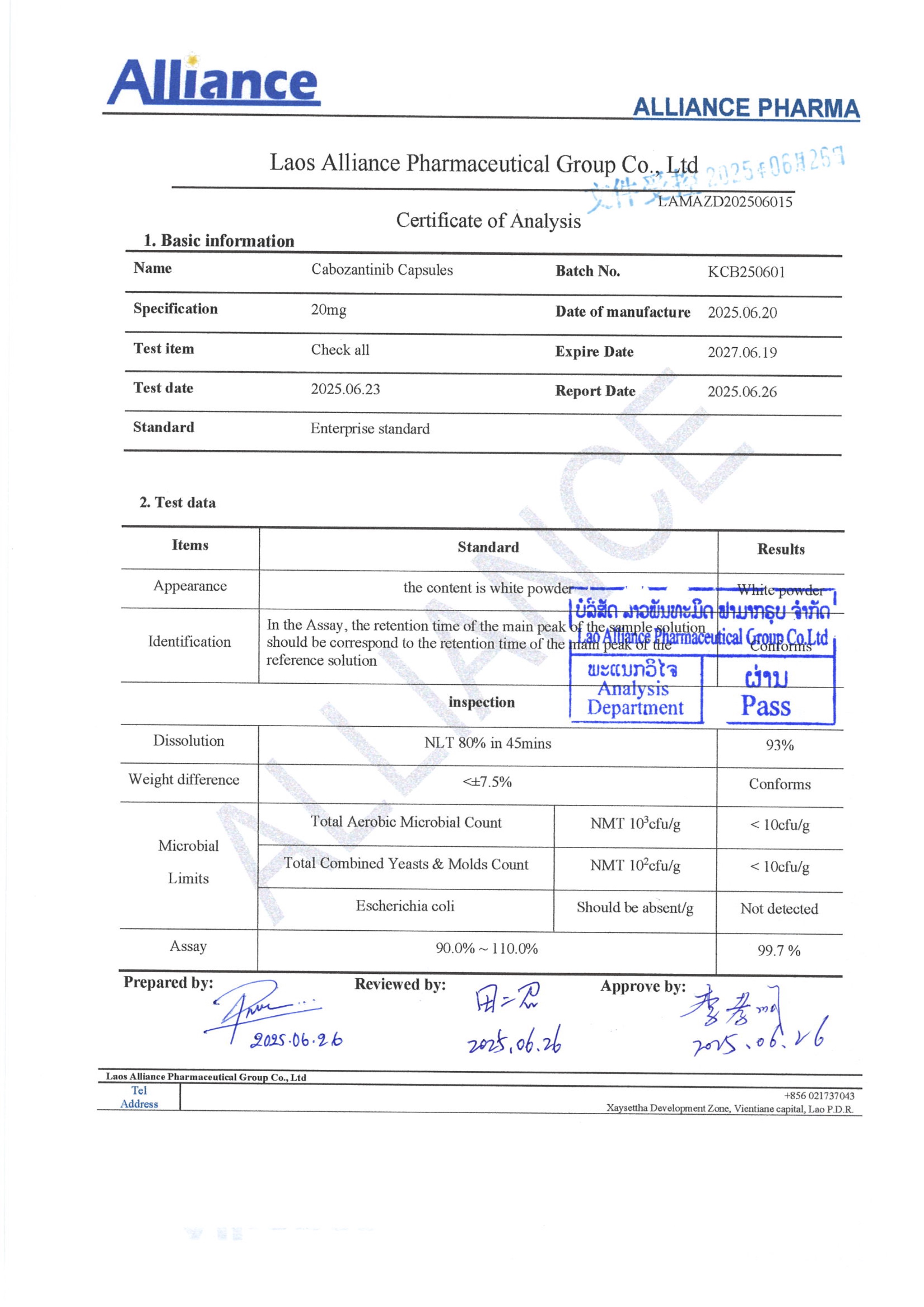
Cabozantinib is a multi-target broad-spectrum anticancer drug developed by Exelixis Inc. (USA) and manufactured and marketed by Lucius (Laos) Pharmaceutical Co. It can inhibit at least 9 targets including MET, VEGFR1/2/3, ROS1, RET, AXL, NTRK, KIT, etc. At present, cabozantinib has proven good therapeutic effects in a variety of solid tumors such as renal cell carcinoma, thyroid carcinoma, hepatocellular carcinoma, soft tissue sarcoma, non-small cell lung cancer, prostate cancer, breast cancer, ovarian cancer, and colorectal cancer, especially showing prominent efficacy in controlling bone metastases. Due to its broad effectiveness against multiple cancers, cabozantinib is known as a
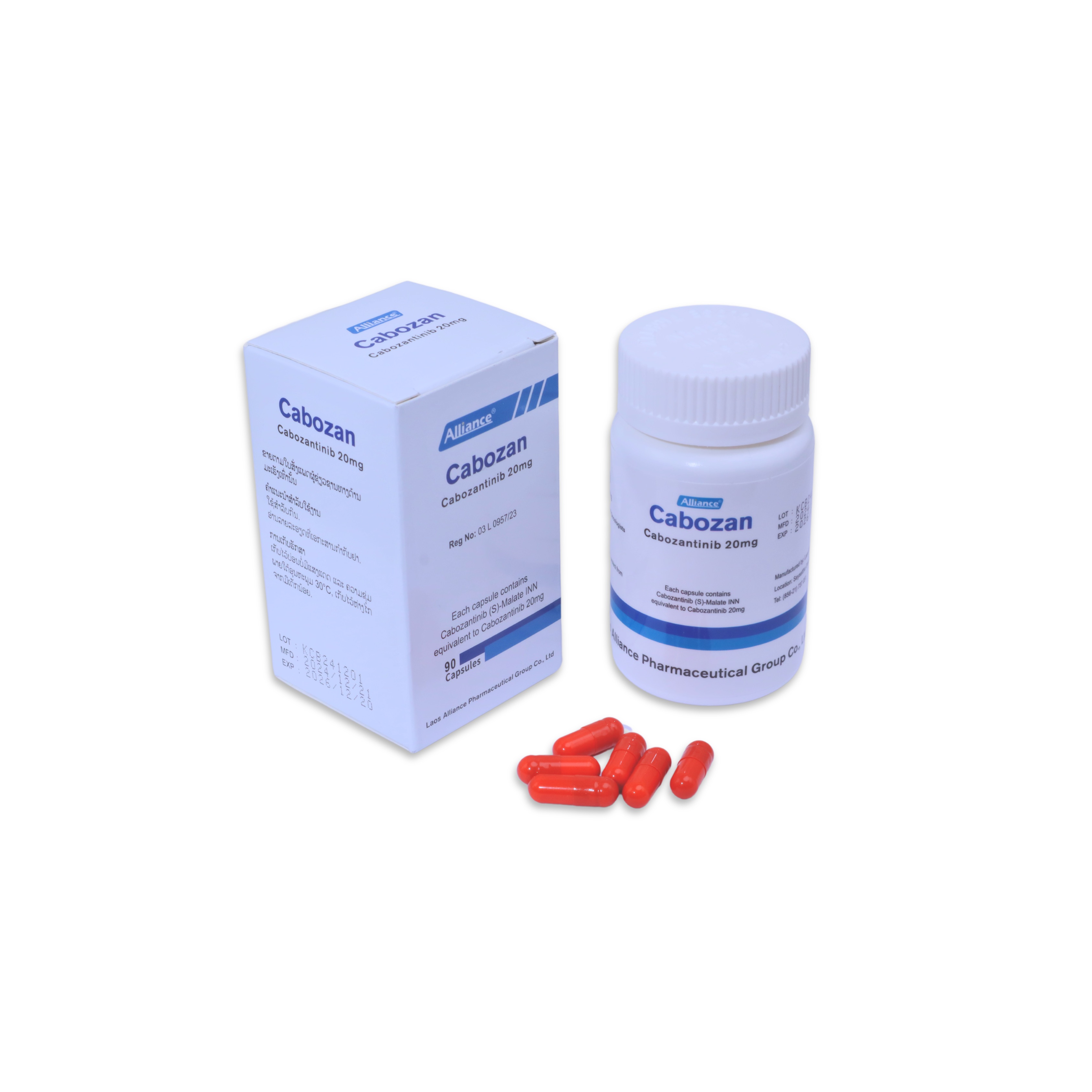
卡博替尼胶囊Cabozantinib 20mg







¥500.00
售价:¥500.00
销量:287 件
Treatment of progressive, unresectable locally advanced or metastatic medullary thyroid carcinoma. Advanced renal cell carcinoma that has failed sunitinib treatment. Advanced non-small cell lung cancer patients with c-Met amplification. Hepatocellular carcinoma patients with resistance to sorafenib. Advanced prostate cancer patients with bone metastases.
20 mg per capsule; 90 capsules per box.
Store at 20°C to 25°C (68°F to 77°F). Short-term excursions between 15°C and 30°C (59°F and 86°F) are permitted for transportation.
Administration Method Oral administration, on an empty stomach. Avoid co-administration with food, i.e., do not eat for 2 hours before dosing and 1 hour after dosing. Swallow the capsules whole. Do not crush cabozantinib tablets. During treatment, do not consume foods or nutritional supplements known to inhibit cytochrome P450 enzymes (e.g., grapefruit, grapefruit juice). Recommended Dose Indication Dose Special Populations Adjustment Medullary Thyroid Carcinoma 140 mg orally once daily Patients with mild to moderate hepatic impairment: starting dose of 80 mg once daily Renal Cell Carcinoma, Hepatocellular Carcinoma 60 mg orally once daily - Prostate Cancer with Bone Metastases 40 mg orally once daily -
Common Adverse Reactions (Incidence > 20%) Diarrhea, stomatitis, palmar-plantar erythrodysesthesia syndrome (PPES), weight loss, decreased appetite, nausea, fatigue, oral pain, hair discoloration, dysgeusia, hypertension, abdominal pain, constipation. Common Laboratory Abnormalities (Incidence > 25%) Elevated AST, elevated ALT, lymphopenia, elevated ALP, hypocalcemia, neutropenia, thrombocytopenia, hypophosphatemia, hyperbilirubinemia. Common Grade 3–4 Adverse Reactions (Incidence > 5%) Diarrhea, palmar-plantar erythrodysesthesia syndrome (PPES), lymphopenia, hypocalcemia, fatigue, hypertension, asthenia, elevated ALT, weight loss, stomatitis, decreased appetite. Common Adverse Reactions Leading to Permanent Discontinuation Hypocalcemia, elevated lipase, palmar-plantar erythrodysesthesia syndrome (PPES), diarrhea, fatigue, hypertension, nausea, pancreatitis, tracheal fistula formation, vomiting.
Cardiac Toxicity Cabozantinib has certain cardiac toxicity, manifested as chest tightness, palpitations, dyspnea, etc. Cardioprotective agents can be taken for prevention, such as Coenzyme Q10. Recommended dosage of Coenzyme Q10: Health maintenance dose for general population: 30–60 mg daily. High-risk groups for cardiovascular diseases (including hypertensive patients): 60–120 mg daily. Patients with heart failure or ischemic heart disease: can be increased to 200–400 mg daily. If the daily dose exceeds 60 mg, it can be taken in 2–3 divided doses. Indications for Permanent Discontinuation Permanent discontinuation is required if severe bleeding, myocardial infarction, cerebral infarction, thromboembolism, hypertensive crisis, nephrotic syndrome, severe hepatitis, osteonecrosis of the jaw, gastrointestinal perforation, or fistula occur during treatment.
Avoid concomitant use with the following drugs during cabozantinib treatment: Strong CYP3A4 Inhibitors Including but not limited to: ketoconazole, itraconazole, clarithromycin, atazanavir, indinavir, nefazodone, nelfinavir, ritonavir, saquinavir, telithromycin, voriconazole. Strong CYP3A4 Inducers Including but not limited to: dexamethasone, phenytoin, carbamazepine, rifampicin, rifabutin, rifapentine, phenobarbital, St. John's wort.
Elderly Patients In phase 3 studies, 41% of patients treated with cabozantinib were aged 65 years and above, and 8% were aged 75 years and above. No differences in safety and efficacy were observed between elderly and younger patients. Patients with Hepatic Impairment Increased exposure to cabozantinib has been observed in patients with mild to moderate hepatic impairment. Reduce the dose of cabozantinib in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. It is not recommended to use cabozantinib in patients with severe hepatic impairment. Patients with Renal Impairment No dose adjustment is required in patients with mild or moderate renal impairment. There is currently no experience with cabozantinib in patients with severe renal impairment.
Pregnant Women: Cabozantinib may cause fetal harm. Pregnant women should avoid using this drug. Females of reproductive potential should use effective contraceptive measures during treatment with cabozantinib and for a certain period after stopping the drug. Lactating Women: It is unknown whether cabozantinib is excreted in human milk. Due to the potential risk of serious adverse reactions in breastfed infants, lactating women should discontinue breastfeeding during treatment with cabozantinib.
评论
添加评论
请登录后发表评论
暂无评论