Brigatinib is a second-generation ALK-targeted therapy. It is currently the first drug that can achieve a progression-free survival (PFS) of more than one year in patients with resistance to crizotinib treatment. The overall response rate of brigatinib is as high as 73% in patients including those previously treated with crizotinib and those untreated with ALK inhibitors.
布加替尼Brigatinib
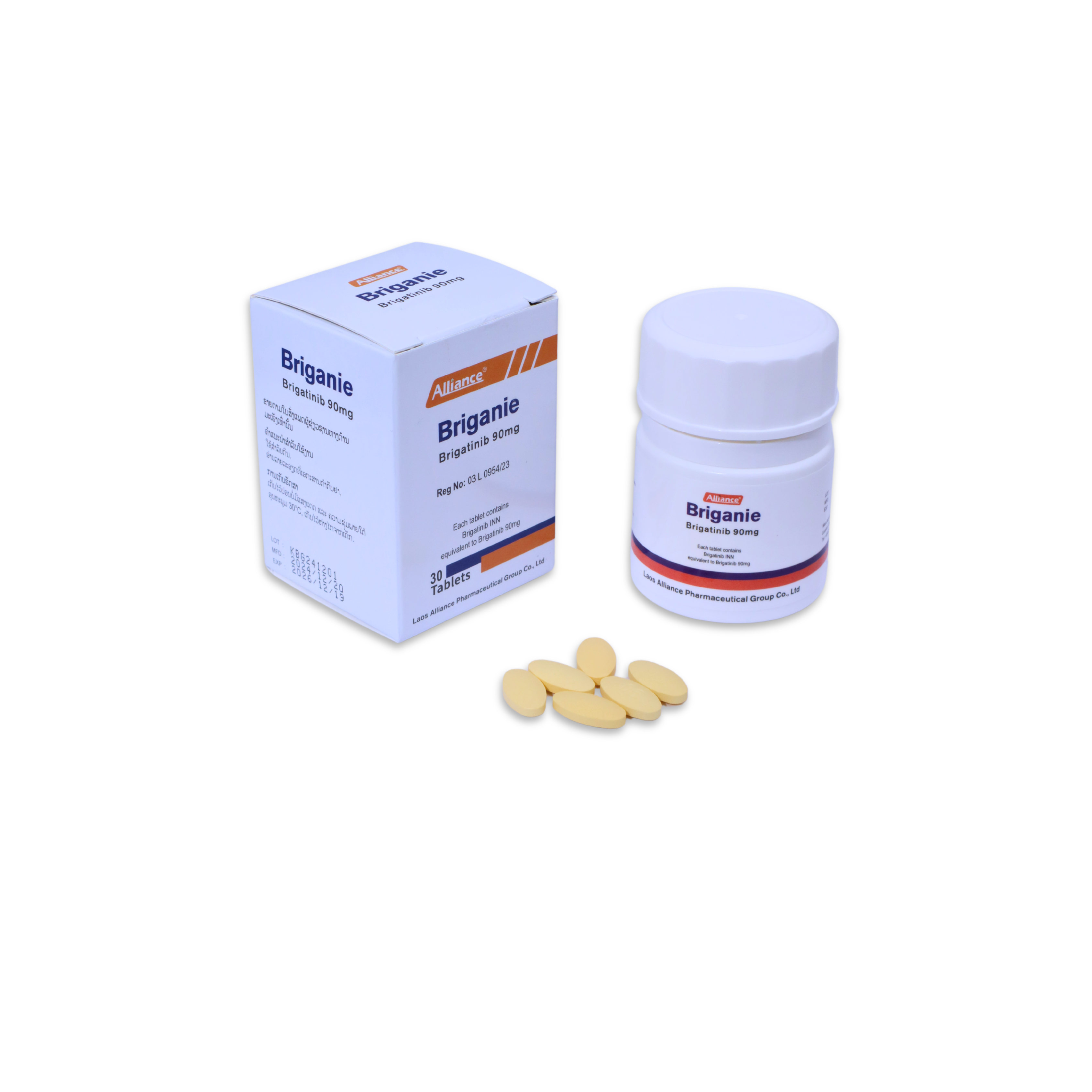

¥500.00
售价:¥500.00
销量:299 件
Brigatinib is a kinase inhibitor indicated for the treatment of patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) who have progressed on or are intolerant to crizotinib.
90 mg per tablet; 30 tablets per bottle.
Store at 20°C to 25°C (68°F to 77°F). Short-term excursions between 15°C and 30°C (59°F and 86°F) are permitted for transportation.
Initiation Phase: 90 mg orally once daily for the first 7 days. Maintenance Phase: If the 90 mg dose is tolerated during the first 7 days, escalate the dose to 180 mg orally once daily.
The most common adverse reactions (≥25%) include nausea, diarrhea, fatigue, cough, and headache.
Interstitial Lung Disease (ILD)/Pneumonitis: Occurs in 9.1% of patients at the recommended dose. Monitor for new or worsening respiratory symptoms, especially during the first week of treatment. Withhold brigatinib in patients with new or worsening respiratory symptoms and promptly evaluate for ILD/pneumonitis. Upon recovery, either reduce the dose or permanently discontinue brigatinib. Hypertension: Monitor blood pressure 2 weeks after the start of treatment and then at least once a month thereafter. Withhold brigatinib in patients with severe hypertension, then reduce the dose or permanently discontinue the drug. Bradycardia: Monitor heart rate and blood pressure regularly during treatment. Withhold brigatinib in symptomatic patients, then reduce the dose or permanently discontinue the drug. Visual Disturbances: Advise patients to report any visual symptoms. Withhold brigatinib and perform an ophthalmologic evaluation, then reduce the dose or permanently discontinue the drug. Creatine Phosphokinase (CPK) Elevation: Monitor CPK levels regularly during treatment. Withhold brigatinib based on the severity of elevation, then resume treatment at the same or reduced dose. Elevated Pancreatic Enzymes: Monitor lipase and amylase levels regularly during treatment. Withhold brigatinib based on the severity of elevation, then resume treatment at the same or reduced dose. Hyperglycemia: Evaluate fasting serum glucose levels before the initiation of brigatinib and regularly during treatment. If hyperglycemia cannot be controlled with optimal medical management, withhold brigatinib, then consider dose reduction or permanent discontinuation based on severity. Embryo-Fetal Toxicity: May cause fetal harm. Advise females of reproductive potential of the potential risk to the fetus and to use effective non-hormonal contraceptive methods.
CYP3A Inhibitors: Avoid concomitant use of brigatinib with strong CYP3A inhibitors. If concomitant use with a strong CYP3A inhibitor is unavoidable, reduce the dose of brigatinib. CYP3A Inducers: Avoid concomitant use of brigatinib with strong CYP3A inducers. CYP3A Substrates: Hormonal contraceptives may be ineffective due to decreased exposure.
评论
添加评论
请登录后发表评论
暂无评论