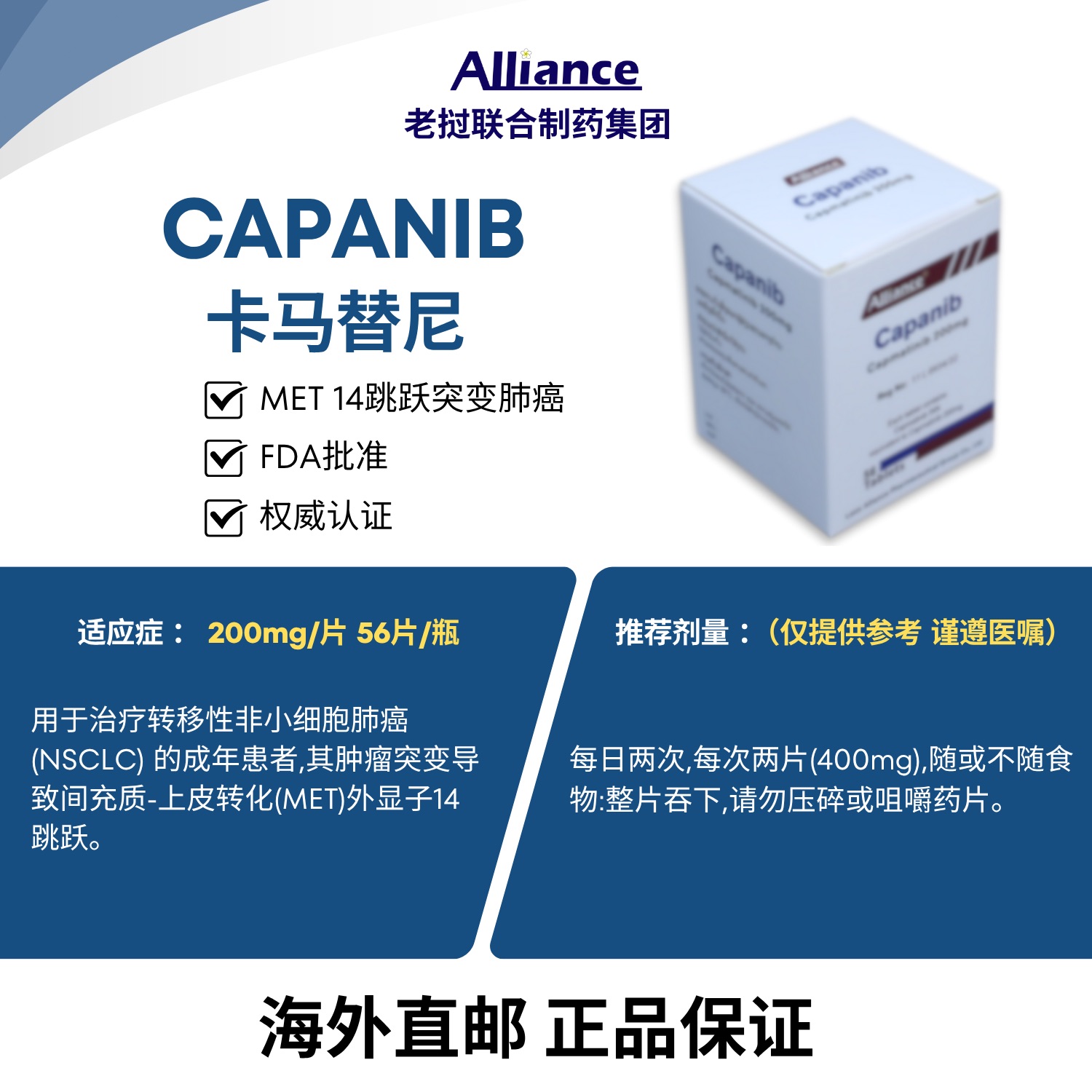
Indicated for the treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) whose tumors harbor mesenchymal-epithelial transition (MET) exon 14 skipping mutations, as detected by an FDA-approved test.
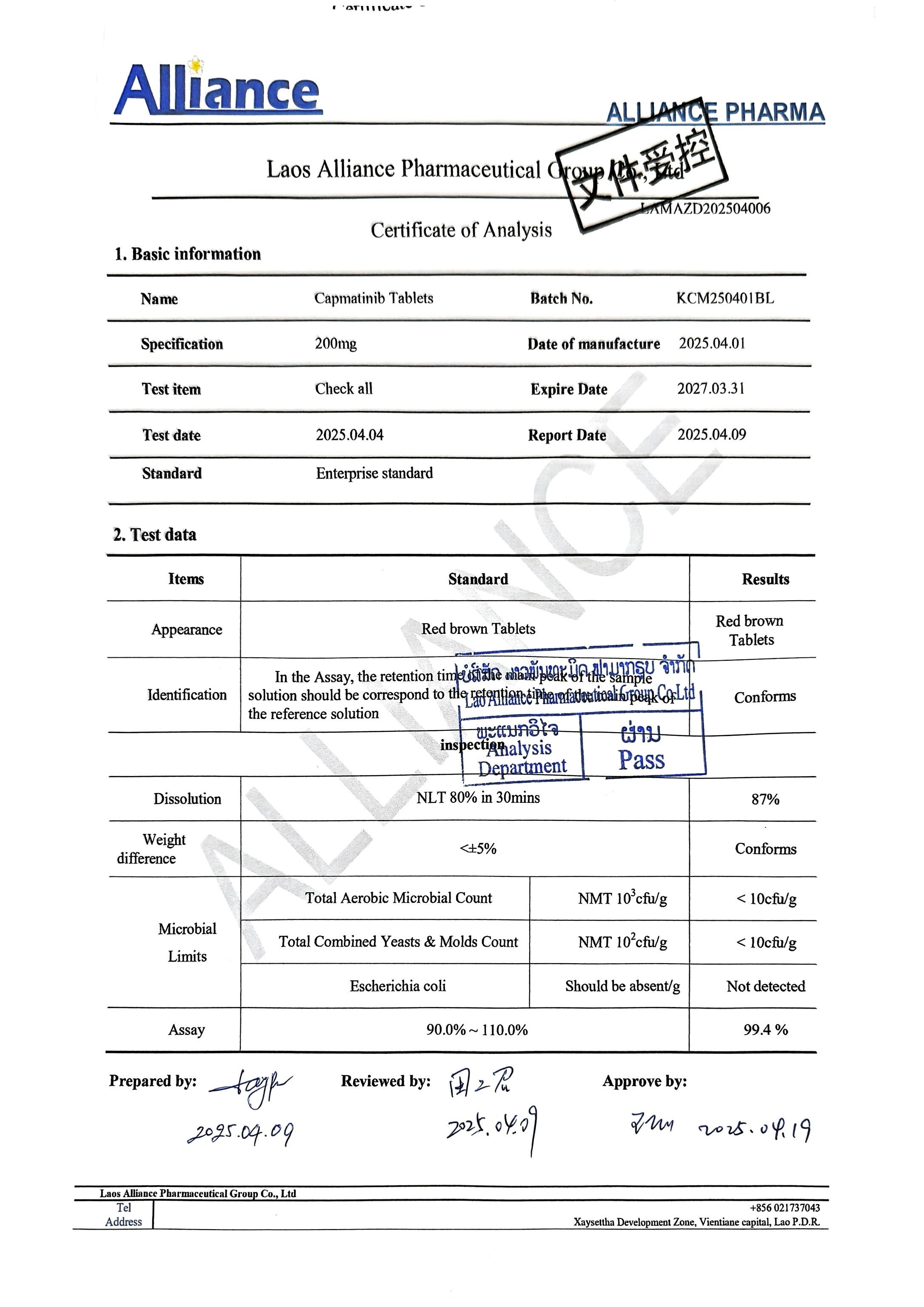
卡马替尼片Capmatinib 200mg







¥2300.00
售价:¥2300.00
销量:99 件
Capmatinib is a kinase inhibitor targeting MET, including mutants generated by exon 14 skipping. MET exon 14 skipping results in the deletion of a regulatory domain in the protein, which reduces its negative regulatory effect and further enhances downstream MET signal transduction. At clinically achievable concentrations, capmatinib can inhibit the growth of cancer cells driven by mutant MET with exon 14 deletion, and exhibits antitumor activity in mouse tumor xenograft models derived from human lung cancer, including tumors with MET exon 14 skipping mutations or MET amplification. Capmatinib inhibits MET phosphorylation triggered by hepatocyte growth factor binding or MET amplification, as well as the phosphorylation of downstream signal proteins mediated by MET, and suppresses the proliferation and survival of MET-dependent cancer cells.
Patient Selection Select patients for capmatinib treatment based on the presence of MET exon 14 skipping mutations in tumor or plasma samples. If no such mutations are detected in plasma samples, tumor tissue testing should be performed whenever possible. Recommended Dose The recommended dose of capmatinib is 400 mg orally twice daily, with or without food. Administration Instructions Swallow the tablets whole. Do not split, crush or chew the tablets. Missed Dose or Vomiting If a dose is missed or vomiting occurs after administration, do not take an extra dose. Instead, take the next scheduled dose at the regular time.
Effects of Other Drugs on Capmatinib Strong CYP3A Inhibitors: During concomitant use of capmatinib with strong CYP3A inhibitors, closely monitor patients for adverse reactions. Strong and Moderate CYP3A Inducers: Avoid concomitant use of capmatinib with strong and moderate CYP3A inducers. Effects of Capmatinib on Other Drugs CYP1A2 Substrates: Concomitant use with capmatinib increases the exposure of CYP1A2 substrates, which may elevate the risk of adverse reactions to these substrates. P-glycoprotein (P-gp) and Breast Cancer Resistance Protein (BCRP) Substrates: Concomitant use with capmatinib increases the exposure of P-gp and BCRP substrates, which may increase the risk of adverse reactions to these substrates. MATE1 and MATE2K Substrates: Concomitant use with capmatinib may increase the exposure of MATE1 and MATE2K substrates, potentially increasing the risk of adverse reactions to these substrates.
Pulmonary or respiratory problems, liver problems, pancreatic problems, swelling of hands and feet, nausea, musculoskeletal pain, fatigue and weakness, vomiting, dyspnea, cough, decreased appetite, changes in certain blood test results.
Interstitial Lung Disease (ILD)/Pneumonitis Discontinue capmatinib immediately in patients with suspected ILD/pneumonitis. Permanently discontinue the drug if no other causes of ILD/pneumonitis are identified. Hepatotoxicity Monitor liver function (including ALT, AST and total bilirubin) before initiating capmatinib. Monitor liver function every 2 weeks during the first 3 months of treatment, then monthly or as clinically indicated thereafter. Increase the frequency of monitoring in patients with elevated transaminases or bilirubin. Suspend treatment, reduce the dose, or permanently discontinue capmatinib based on the severity of adverse reactions. Pancreatic Toxicity Monitor amylase and lipase levels at baseline and periodically during capmatinib treatment. Temporarily suspend treatment, reduce the dose, or permanently discontinue capmatinib based on the severity of adverse reactions. Photosensitivity Risk Advise patients to limit direct exposure to ultraviolet (UV) light during capmatinib treatment. Embryo-Fetal Toxicity Based on animal study results and its mechanism of action, capmatinib may cause fetal harm when administered to pregnant women.
Store in a cool, dry place below 25℃, protected from light. Keep out of the reach of children.
评论
添加评论
请登录后发表评论
暂无评论