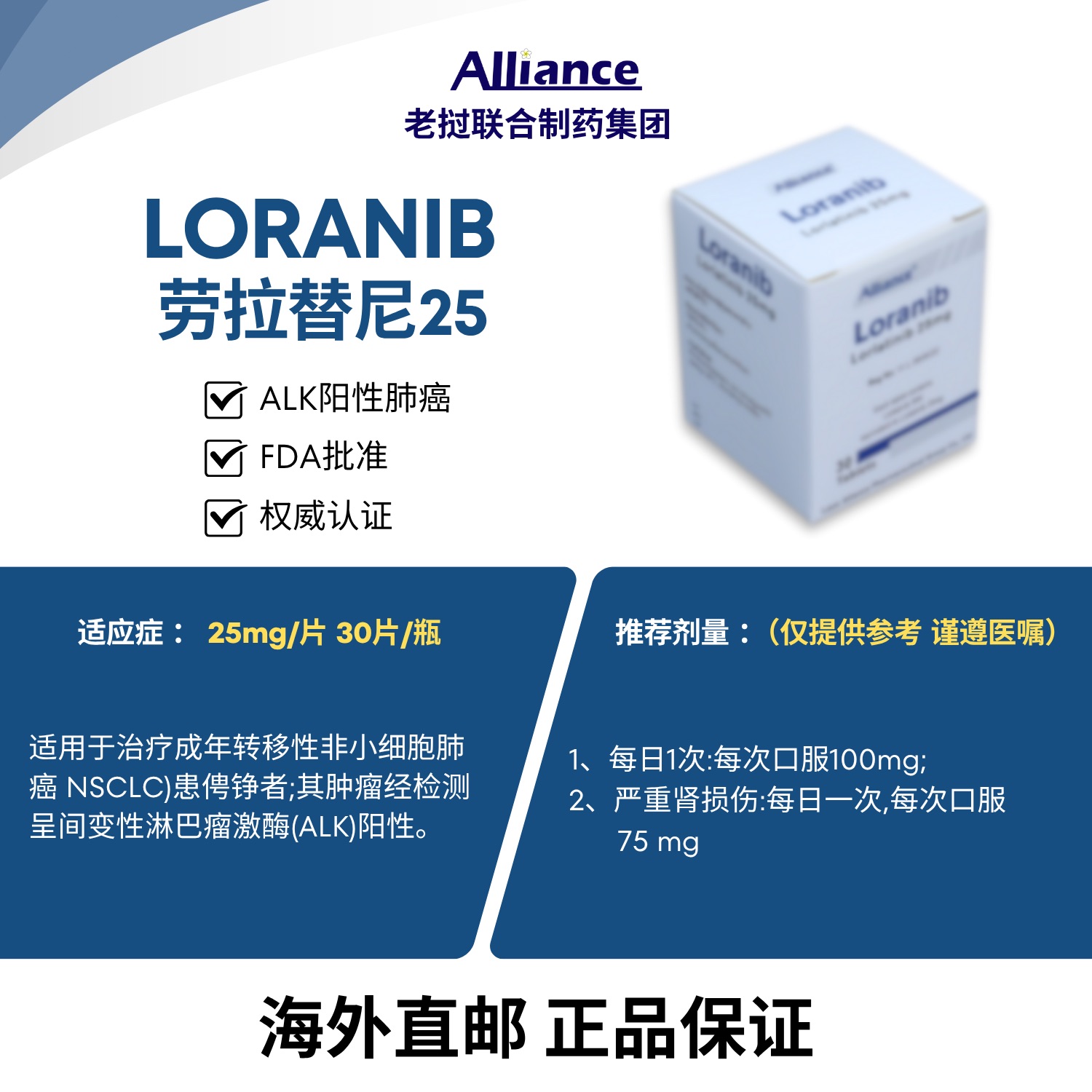
Lorlatinib as monotherapy is indicated for the treatment of adult patients with anaplastic lymphoma kinase (ALK)-positive advanced non-small cell lung cancer (NSCLC) who have progressed after: Alectinib or ceritinib as the first-line ALK inhibitor therapy; Crizotinib and at least one other ALK inhibitor therapy.
劳拉替尼片Lorlatinib 25mg




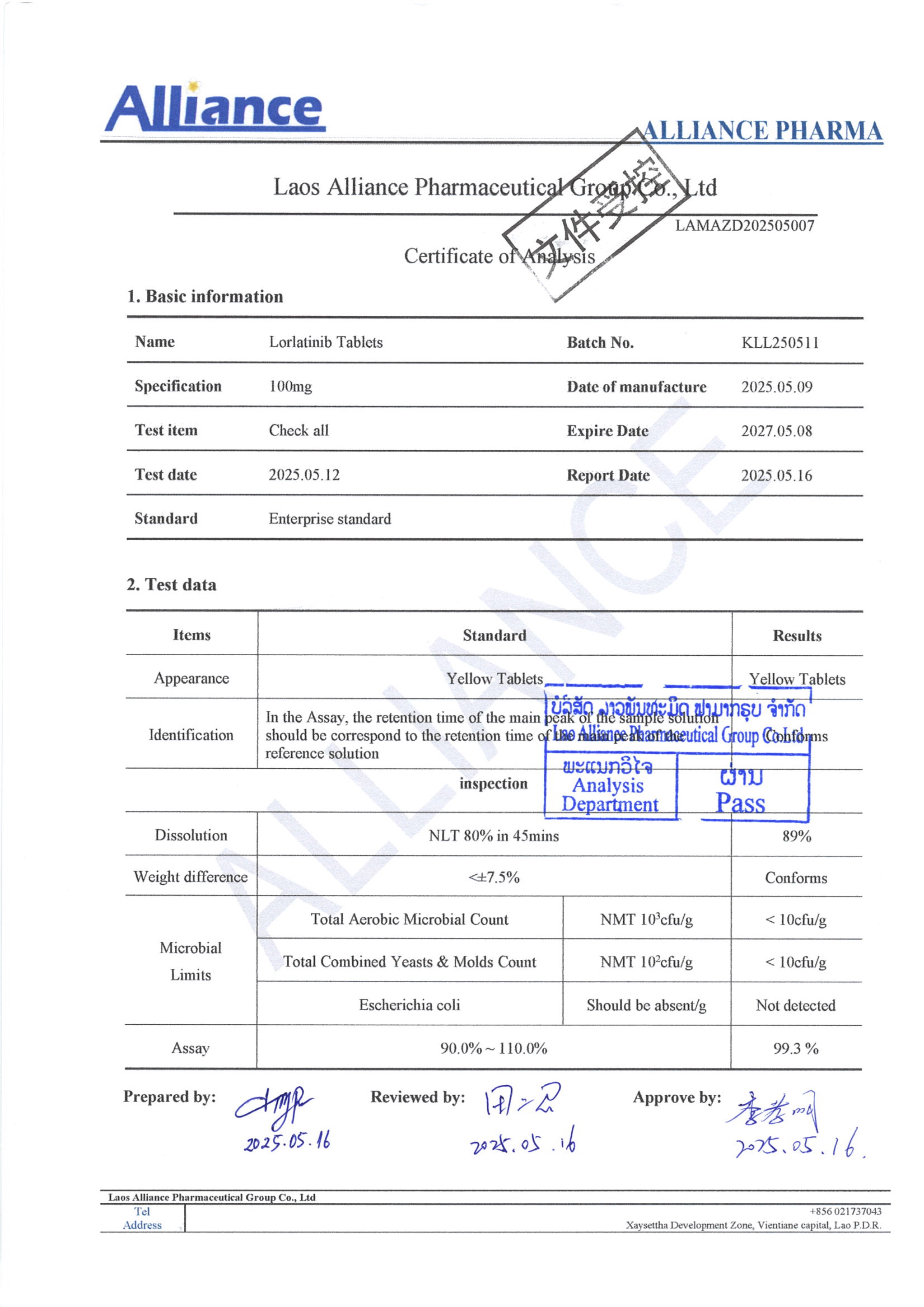


¥980.00
售价:¥980.00
销量:680 件
Lorlatinib is a kinase inhibitor. In vitro studies have shown that it exhibits activity against ALK, ROS1, TYK1, FER, FPS, TRKA, TRKB, TRKC, FAK, FAK2 and ACK. Lorlatinib has demonstrated activity against multiple ALK enzyme mutants in vitro, including some mutations detected in tumors that progressed after treatment with crizotinib and other ALK inhibitors. Antitumor activity was observed in mice subcutaneously implanted with tumors carrying EML4 fusion genes (fusion genes containing ALK variant 1 or ALK mutations, including G1202R and I1171T mutations detected in tumors progressing after ALK inhibitor treatment) following administration of lorlatinib. In addition, lorlatinib showed antitumor activity and prolonged the survival of mice intracranially implanted with EML4-ALK-driven tumor cell lines. In in vivo models, the overall antitumor activity of lorlatinib was dose-dependent and correlated with the inhibition of ALK phosphorylation.
Recommended Dose: 100 mg orally once daily. Treatment Duration: Continue treatment as long as the patient derives clinical benefit and no unacceptable toxicity occurs. Missed Dose: If a dose is missed, take it as soon as it is remembered, unless the next scheduled dose is due within 4 hours. Do not take two doses to make up for a missed dose. Dose Reduction: Dose interruption or reduction may be required based on individual safety and tolerability. The dose reduction levels are summarized as follows: First dose reduction: 75 mg orally once daily. Second dose reduction: 50 mg orally once daily. Permanently discontinue lorlatinib if the patient is unable to tolerate 50 mg orally once daily.
CYP3A4/5 Substrates Lorlatinib is a time-dependent inhibitor and inducer of CYP3A4/5 in vitro, and can activate the human pregnane X receptor (PXR), with a net in vivo effect of induction. When patients take lorlatinib and midazolam concomitantly, the oral AUC of midazolam is decreased, indicating that lorlatinib is an inducer of CYP3A4/5. Oral administration of 150 mg lorlatinib once daily for 15 days reduced the AUCinf and Cmax of a single oral 2 mg dose of midazolam (a sensitive CYP3A substrate) by 61% and 50%, respectively; thus, lorlatinib is a moderate-strength CYP3A inducer. Therefore, concomitant use of lorlatinib with CYP3A4/5 substrates with a narrow therapeutic index should be avoided, including but not limited to alfentanil, cyclosporine, dihydroergotamine, ergotamine, fentanyl, hormonal contraceptives, pimozide, quinidine, sirolimus and tacrolimus, as lorlatinib may decrease the concentrations of these drugs. Other CYP Inhibition and Induction (In Vitro) Lorlatinib may have the potential to inhibit CYP2C9. Lorlatinib is an inducer of CYP2B6 and can activate the human constitutive androstane receptor (CAR). Concomitant use of lorlatinib with CYP2B6 substrates (e.g., bupropion, efavirenz) may result in decreased plasma concentrations of CYP2B6 substrates. The possibility of drug interactions caused by lorlatinib through induction of CYP1A2 is low. UGT Inhibition (In Vitro) Lorlatinib may have the potential to inhibit UGT1A1. Drug Transporters (In Vitro) At clinically relevant concentrations, lorlatinib may have the potential to inhibit P-glycoprotein (P-gp, systemic and gastrointestinal tract), BCRP (gastrointestinal tract), OATP1B1, OATP1B3, OCT1, MATE1 and OAT3.
The most commonly reported adverse reactions include: Hypercholesterolemia (84.4%) Hypertriglyceridemia (67.1%) Edema (54.6%) Peripheral neuropathy (47.8%) Cognitive dysfunction (28.8%) Fatigue (28.1%) Weight gain (26.4%) Mood disorders (22.7%) Dose reduction due to adverse reactions occurred in 23.4% of patients treated with lorlatinib. The most common adverse reactions leading to dose reduction were edema and peripheral neuropathy. Permanent discontinuation due to adverse reactions occurred in 3.1% of patients, with cognitive dysfunction being the most common cause.
Hyperlipidemia Lorlatinib use is associated with elevated serum cholesterol and triglyceride levels. The median time to onset of severe elevations in serum cholesterol and triglycerides was 20 days (range: 4 to 518 days) and 127 days (range: 15 to 358 days), respectively. Monitor serum cholesterol and triglyceride levels before initiation of lorlatinib, at 2, 4 and 8 weeks after initiation, and periodically thereafter. Initiate or increase the dose of lipid-lowering agents as necessary. Central Nervous System (CNS) Effects CNS effects including changes in cognitive function, mood or speech have been observed in patients taking lorlatinib. Dose adjustment or discontinuation may be required for patients with persistent or severe CNS effects. Atrioventricular (AV) Block The lorlatinib study population excluded patients with second- or third-degree AV block (unless paced) or any AV block with a PR interval > 220 ms. PR interval prolongation and AV block have been reported in patients receiving lorlatinib. Monitor electrocardiogram (ECG) before initiation of lorlatinib, especially in patients with predisposing factors for clinically significant cardiac events. Dose adjustment may be required for patients who develop AV block. Decreased Left Ventricular Ejection Fraction (LVEF) Decreased LVEF has been reported in patients receiving lorlatinib who had at least one follow-up LVEF assessment. Based on available clinical study data, a causal relationship between lorlatinib and changes in myocardial contractility cannot be determined. Consider cardiac monitoring, including baseline and on-treatment LVEF assessments, in patients with pre-existing heart disease and those at risk of LVEF impairment. Cardiac monitoring including LVEF assessment should be considered in patients who develop relevant cardiac signs/symptoms during treatment. Elevated Lipase and Amylase Elevated lipase and/or amylase levels have been reported in patients receiving lorlatinib. The median time to onset of elevated serum lipase and amylase was 70 days (range: 7 to 696 days) and 41 days (range: 7 to 489 days), respectively. Due to the combination of hypertriglyceridemia and/or potential underlying mechanisms, the risk of pancreatitis should be considered in patients receiving lorlatinib. Monitor lipase and amylase levels before initiation of lorlatinib and periodically as clinically indicated during treatment. Interstitial Lung Disease (ILD)/Pneumonitis Severe or life-threatening pulmonary adverse reactions consistent with ILD/pneumonitis have occurred after lorlatinib administration. Any patient with worsening respiratory symptoms suggestive of ILD/pneumonitis (e.g., dyspnea, cough, fever) should be promptly evaluated for ILD/pneumonitis. Interrupt and/or permanently discontinue lorlatinib based on the severity of the reaction. Drug Interactions Strong CYP3A4/5 Inducers: In a study of healthy volunteers, concomitant use of lorlatinib with rifampin (a strong CYP3A4/5 inducer) resulted in elevated alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels, with no elevation in total bilirubin or alkaline phosphatase. Concomitant use of lorlatinib with strong CYP3A4/5 inducers is contraindicated. Concomitant use with moderate CYP3A4/5 inducers should be avoided whenever possible, as they may also decrease lorlatinib plasma concentrations. CYP3A4/5 Substrates with Narrow Therapeutic Index: Avoid concomitant use, as lorlatinib may decrease their concentrations. Strong CYP3A4/5 Inhibitors: Itraconazole (a strong CYP3A4/5 inhibitor) administered orally at 200 mg once daily for 5 days increased the mean AUC and Cmax of a single 100 mg oral dose of lorlatinib by 42% and 24%, respectively, in healthy volunteers. Concomitant use of lorlatinib with strong CYP3A4/5 inhibitors (e.g., boceprevir, cobicistat, itraconazole, ketoconazole, posaconazole, telithromycin, voriconazole, ritonavir, paritaprevir co-administered with ritonavir and ombitasvir and/or dasabuvir, and ritonavir co-administered with elvitegravir, indinavir, lopinavir or tipranavir) may increase lorlatinib plasma concentrations. Grapefruit products may also increase lorlatinib plasma concentrations and should be avoided. Consider using alternative drugs with less CYP3A4/5 inhibitory activity. If concomitant use of a strong CYP3A4/5 inhibitor is unavoidable, dose reduction of lorlatinib is recommended. Fertility and Pregnancy Male Patients: Men with female partners of reproductive potential must use effective contraception (including condoms) during treatment with lorlatinib and for at least 14 weeks after the last dose. Men with pregnant partners must use condoms. Male fertility may be affected during lorlatinib treatment. Male patients should consult about effective fertility preservation methods before treatment. Female Patients: Women of reproductive potential should avoid pregnancy during treatment with lorlatinib. Since lorlatinib may render hormonal contraceptives ineffective, female patients must use highly effective non-hormonal contraceptive methods during treatment. If hormonal contraceptives must be used, condoms must also be used concomitantly. Effective contraception must be continued for at least 35 days after treatment completion. The effect of lorlatinib on female fertility is unknown. Dietary Sodium Each 25 mg or 100 mg tablet of this product contains less than 1 millimole (23 mg) of sodium. Patients on a low-sodium diet should be informed that this product is essentially
Store below 30°C. Protect from moisture and light. Keep lorlatinib out of sight and reach of children.
评论
添加评论
请登录后发表评论
暂无评论