Tirzepatide is indicated as an adjunct to diet and exercise to improve glycemic control in adult patients with type 2 diabetes mellitus. Usage Restrictions Not studied in patients with a history of pancreatitis. Not indicated for patients with type 1 diabetes mellitus.
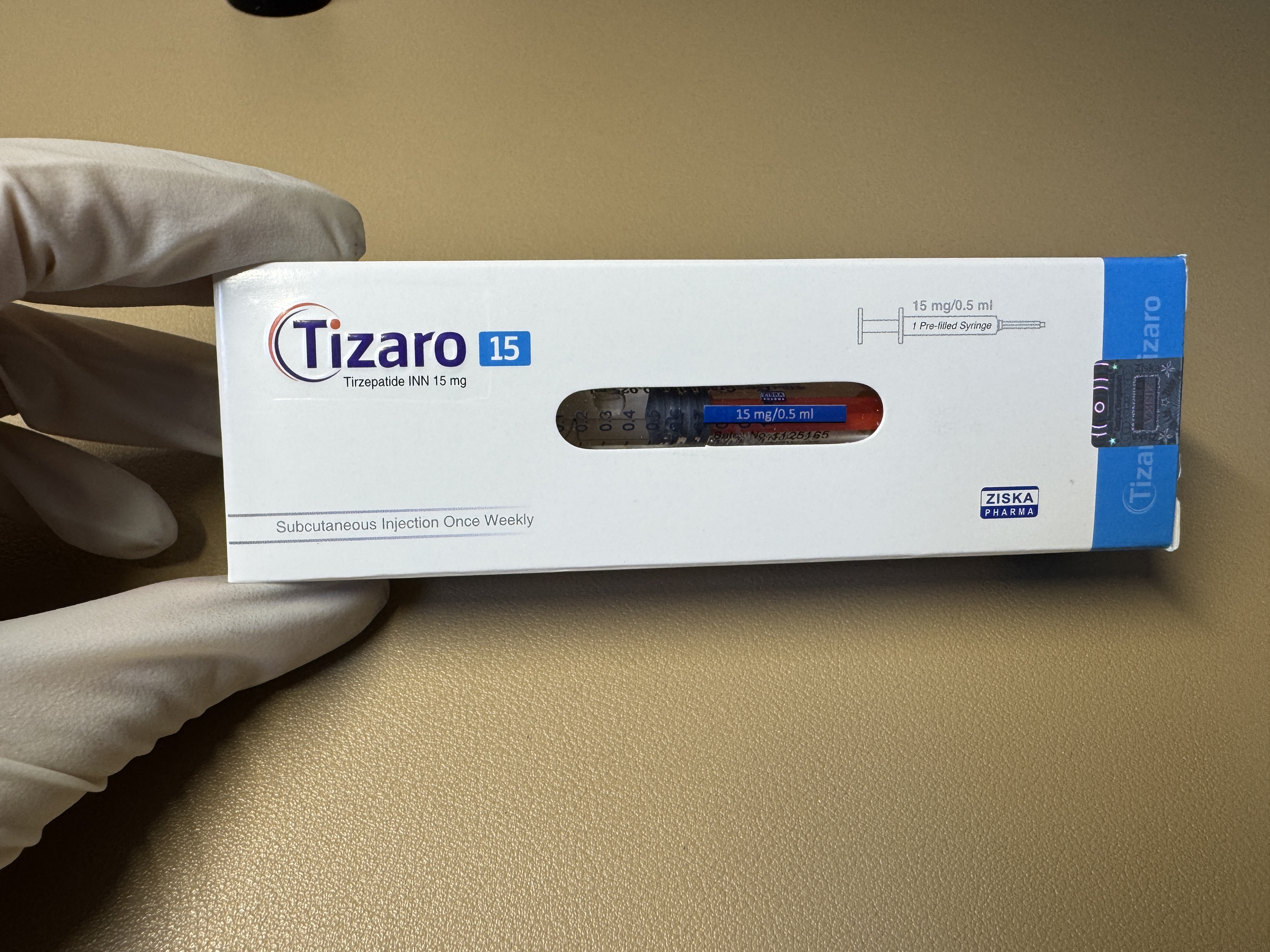
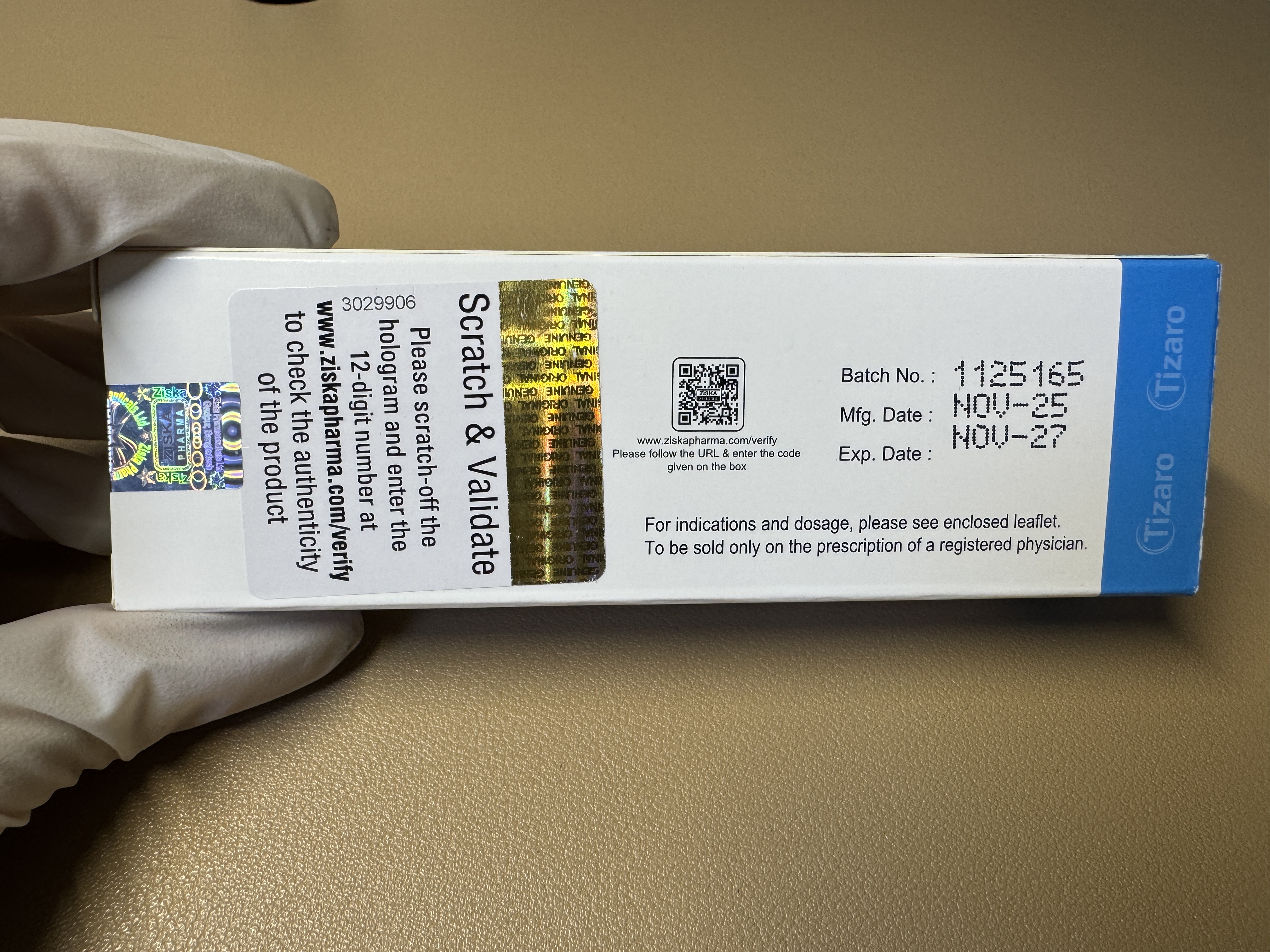
替西帕肽tizaro-15


¥400.00
售价:¥400.00
销量:268 件
Tirzepatide is a glucose-dependent insulinotropic polypeptide (GIP) receptor and glucagon-like peptide-1 (GLP-1) receptor agonist. It is a modified 39-amino acid peptide with a C20 fatty acid moiety that binds to albumin and prolongs its half-life. It selectively binds to and activates GIP and GLP-1 receptors, which are the targets of endogenous GIP and GLP-1. It enhances the first and second phases of insulin secretion in a glucose-dependent manner and reduces glucagon levels.
Administration Route Subcutaneous injection only. Dose Instructions Recommended Initial Dose: 2.5 mg subcutaneously once weekly. The 2.5 mg dose is for treatment initiation only and not for glycemic control. Dose Escalation: Maintain the current dose for at least 4 weeks before increasing by 2.5 mg each time. After 4 weeks, escalate the dose to 5 mg once weekly. Further escalation may be considered for additional glycemic control. Maximum Dose: 15 mg subcutaneously once weekly. Missed Dose: If a dose is missed, administer it as soon as possible within 4 days (96 hours) after the missed dose. If more than 4 days have passed, skip the missed dose and take the next dose on the originally scheduled date. Resume the regular once-weekly dosing schedule thereafter. Dosing Day Adjustment: The day of weekly administration may be changed if necessary, provided that the interval between two doses is at least 3 days (72 hours). Preparation and Handling The needle shield of the pre-filled syringe contains dry natural rubber (a latex derivative), which may cause allergic reactions. Do not freeze. Do not use frozen tirzepatide. Protect the pre-filled syringe from light. Inspect visually for particulate matter and discoloration before administration. Do not use if particulates or discoloration are observed. Handle with care. Do not use the pre-filled syringe if it has been dropped on a hard surface. Do not dilute tirzepatide or administer it concomitantly with other drug solutions. Administration Instructions May be administered at any time of the day, with or without meals. Inject subcutaneously into the abdomen, thigh, or upper arm. Rotate injection sites with each dose. If used concomitantly with insulin, administer as separate injections. Do not mix the two drugs. Tirzepatide and insulin may be injected in the same body region, but the injection sites should not be adjacent.
For concomitant use with insulin secretagogues (e.g., sulfonylureas) or insulin, consider reducing the dose of the insulin secretagogue or insulin to minimize the risk of hypoglycemia. Tirzepatide delays gastric emptying, which may affect the absorption of concomitantly administered oral medications. Monitor patients taking oral drugs with efficacy dependent on threshold concentrations or with a narrow therapeutic index (e.g., warfarin). For patients using oral hormonal contraceptives, recommend switching to non-oral contraceptive methods or adding a barrier contraceptive within 4 weeks after initiating tirzepatide and within 4 weeks after each dose escalation. Non-oral hormonal contraceptives are not affected.
The most common adverse reactions are nausea, diarrhea, decreased appetite, vomiting, constipation, dyspepsia, and abdominal pain.
Patients with known hypersensitivity to tirzepatide or any of its components. Patients with a personal or family history of medullary thyroid carcinoma (MTC). Patients with type 2 multiple endocrine neoplasia syndrome (MEN 2).
Risk of Medullary Thyroid Carcinoma (MTC): Inform patients of the potential risk of MTC and advise them of the symptoms of thyroid tumors. Pancreatitis: Closely monitor patients for signs and symptoms of pancreatitis after initiation of treatment. If pancreatitis is suspected, discontinue tirzepatide and initiate appropriate treatment. Hypoglycemia Risk Reduction: The risk of hypoglycemia can be reduced by lowering the dose of sulfonylureas (or other concomitantly used insulin secretagogues) or insulin. Allergic Reactions: Discontinue tirzepatide immediately if an allergic reaction occurs; initiate standard treatment and continue monitoring until signs and symptoms resolve. Renal Function Monitoring: For patients with renal impairment who report severe gastrointestinal adverse reactions, monitor renal function when initiating treatment or escalating the dose. Diabetic Retinopathy: Monitor patients with a history of diabetic retinopathy for disease progression. Cholelithiasis: If gallstones are suspected, perform diagnostic imaging of the gallbladder and conduct appropriate clinical follow-up.
Keep out of the reach of children. Store in a refrigerator at 2℃ to 8℃. Do not freeze and protect from light. Do not use frozen medication. This product is available only with a registered physician's prescription.
评论
添加评论
请登录后发表评论
暂无评论