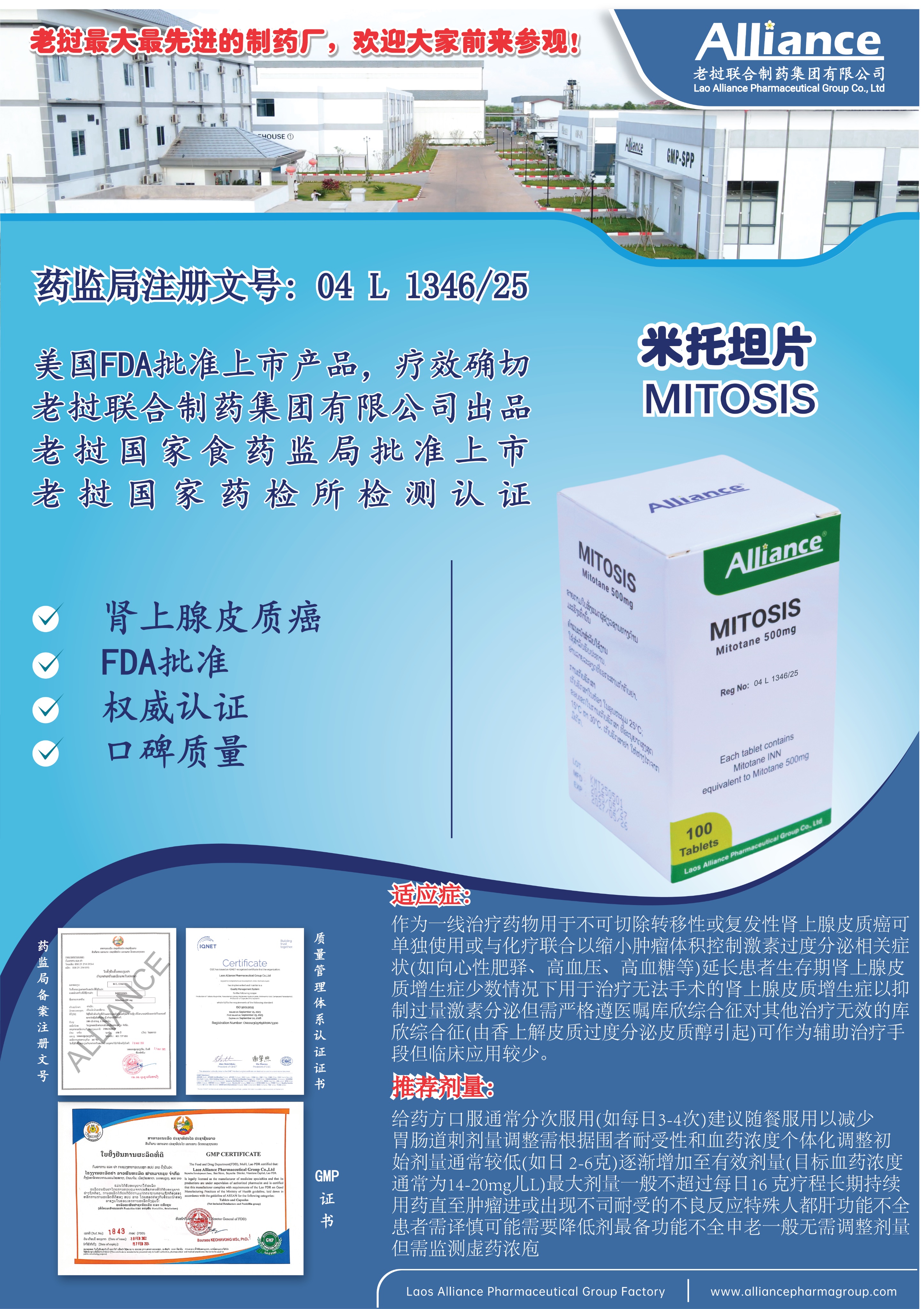
MITOTANE is indicated for the treatment of patients with inoperable functional or non-functional adrenocortical carcinoma.
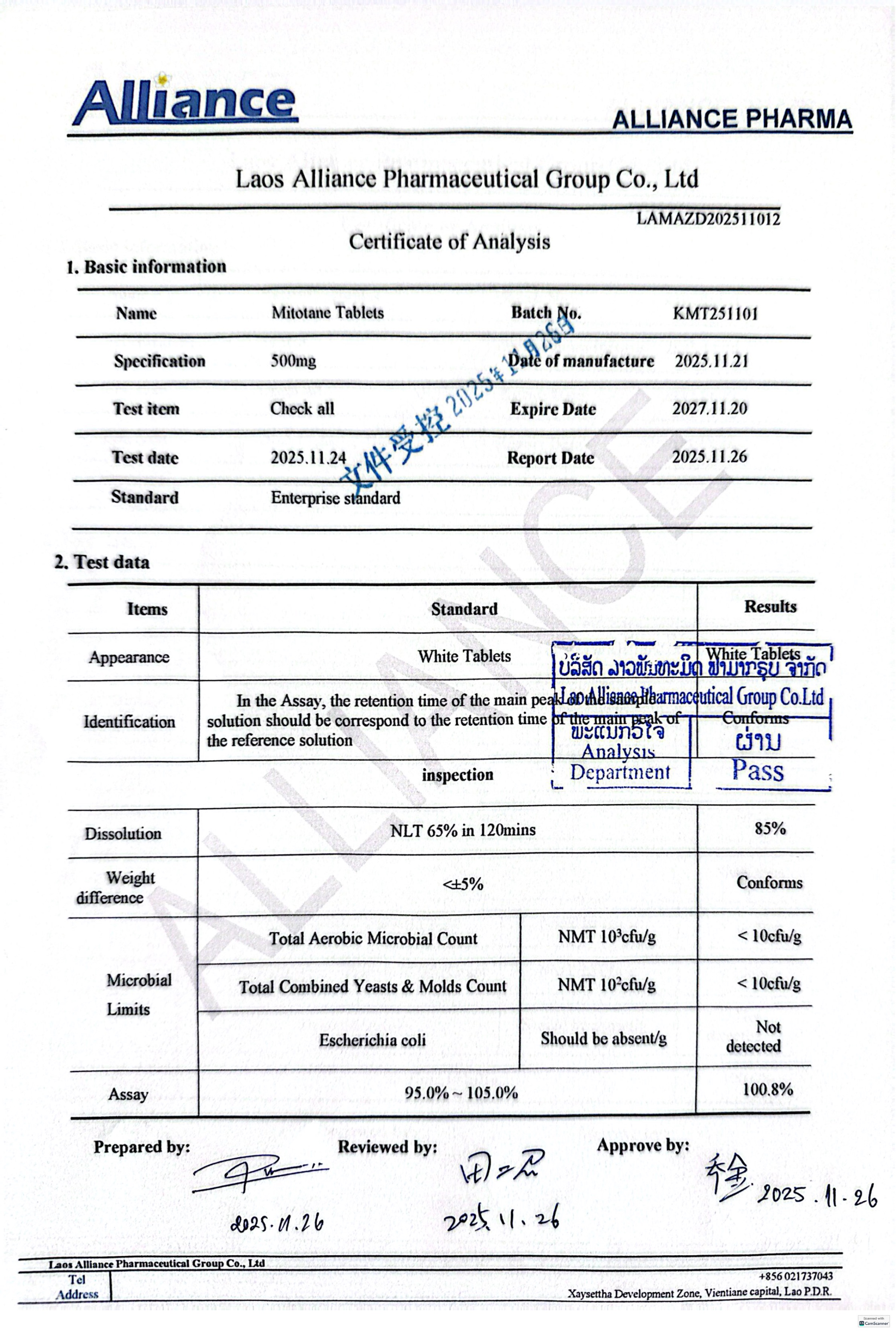
米托坦片Mitotane 500mg




¥1680.00
售价:¥1680.00
销量:329 件
Recommended Initial Dose: 2 g to 6 g orally daily, divided into 3 to 4 administrations. Gradually increase the dose to achieve a plasma concentration of 14 to 20 mg/L, or adjust within the range tolerated by the patient. Special Precautions: MITOTANE is a cytotoxic drug; follow the relevant special handling and disposal procedures.
Adrenal Crisis: Patients receiving MITOTANE may develop adrenal crisis in cases of shock or severe trauma, and their response to shock may be attenuated. Hydrocortisone therapy should be administered, signs of worsening shock should be monitored, and MITOTANE treatment should be suspended until the patient recovers. Central Nervous System (CNS) Toxicity: MITOTANE treatment may cause CNS toxicity, including somnolence, lethargy and dizziness. The incidence of toxicity is higher when the plasma concentration of mitotane exceeds 20 mcg/mL. Adrenal Insufficiency: MITOTANE treatment may lead to adrenal insufficiency. Administer hormone replacement therapy as clinically indicated, and optimize the therapy by measuring free cortisol and adrenocorticotropic hormone (ACTH) levels. Pregnancy Warning: MITOTANE may cause fetal harm. Patients exposed to mitotane during pregnancy may experience abnormal pregnancy outcomes such as preterm birth or early miscarriage. Advise pregnant women of the potential risk to the fetus, and recommend that females of reproductive potential use effective contraceptive measures during treatment and for as long as mitotane plasma levels are detectable after discontinuation. Ovarian Cysts: Bilateral and multiple ovarian cysts may occur in females of reproductive potential receiving MITOTANE treatment. These cysts may lead to complications such as adnexal torsion and cyst rupture with hemorrhage. Improvement may be observed after discontinuation of MITOTANE in some cases. Advise female patients to seek medical attention promptly if they develop gynecological symptoms such as vaginal bleeding and/or pelvic pain.
Common adverse reactions include: anorexia, nausea, vomiting and diarrhea; depression, dizziness or vertigo; and rash.
100 tablets per bottle.
Store at 25°C; short-term excursions between 15°C and 30°C are permitted. Keep out of the reach of children.
评论
添加评论
请登录后发表评论
暂无评论