Clinically approved indications cover a variety of solid tumors, specifically including: Maintenance treatment for adult patients with platinum-sensitive recurrent epithelial ovarian cancer, fallopian tube cancer, or primary peritoneal cancer, who have achieved complete or partial remission after platinum-containing chemotherapy. Monotherapy for adult patients with advanced ovarian cancer, fallopian tube cancer, or primary peritoneal cancer harboring BRCA1/2 gene mutations, who have received three or more lines of chemotherapy. Monotherapy for adult patients with HER2-negative metastatic breast cancer harboring germline BRCA gene mutations, who have previously received chemotherapy (hormone receptor-positive patients with disease progression after endocrine therapy should receive endocrine therapy first). Maintenance treatment for adult patients with metastatic pancreatic cancer harboring germline or somatic BRCA gene mutations, who have no disease progression after first-line platinum-containing chemotherapy. Treatment for adult patients with metastatic castration-resistant prostate cancer harboring homologous recombination repair (HRR) gene mutations.
奥拉帕利片Olaparib 150mg


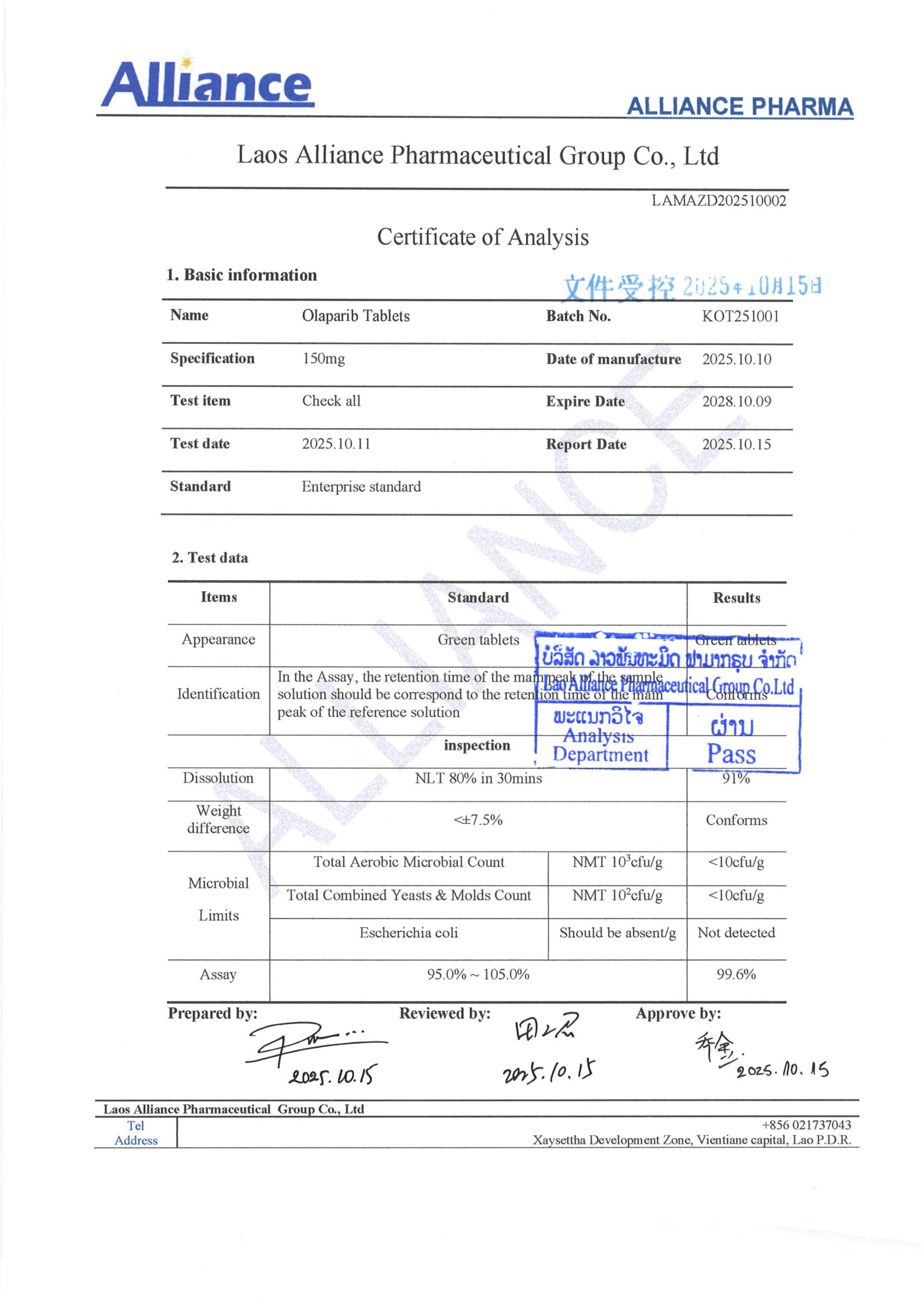

¥1200.00
售价:¥1200.00
销量:226 件
Recommended Dose 300 mg orally twice daily, with or without food. Olaparib monotherapy: Patients should initiate treatment with this product within 8 weeks after the completion of platinum-containing chemotherapy. Olaparib in combination with bevacizumab: Patients should initiate combination therapy within 3–9 weeks after the completion of first-line platinum-containing chemotherapy combined with bevacizumab. The dose of bevacizumab is 15 mg/kg once every 3 weeks; please refer to the package insert of bevacizumab injection for details.
PARP enzymes are involved in the repair of DNA single-strand breaks. When tumor cells have HRR-related gene mutations such as BRCA1/2, their DNA double-strand break repair pathway is defective. Olaparib can inhibit PARP enzyme activity, block DNA single-strand break repair, leading to the continuous accumulation of DNA damage in tumor cells, and ultimately trigger tumor cell apoptosis. This killing method of
Common Adverse Reactions Nausea, vomiting, diarrhea, dyspepsia, fatigue, asthenia, anemia, thrombocytopenia, neutropenia, alopecia, headache, etc., mostly mild to moderate in severity. Severe Adverse Reactions Myelodysplastic Syndrome/Acute Myeloid Leukemia (MDS/AML): Routine blood tests should be monitored regularly during medication. If persistent cytopenia occurs, patients need to seek medical attention promptly for further evaluation. Pneumonitis: Manifested as cough, dyspnea, etc. Discontinue the drug immediately and provide symptomatic treatment when relevant symptoms occur. Fetal Toxicity: Potential harm to the fetus.
Avoid concomitant use with potent CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, clarithromycin), which can significantly increase the blood concentration of olaparib and increase the risk of adverse reactions. If concomitant use is unavoidable, reduce the dose of olaparib by half. Avoid concomitant use with potent CYP3A4 inducers (e.g., rifampicin, carbamazepine, phenytoin sodium), which can decrease the blood concentration of olaparib and affect efficacy.
Hepatic Impairment No dose adjustment is required for patients with mild hepatic impairment. The recommended dose for patients with moderate hepatic impairment is 200 mg orally twice daily. Use with caution in patients with severe hepatic impairment; no safe dose data is available. Renal Impairment No dose adjustment is required for patients with mild to moderate renal impairment. The recommended dose for patients with severe renal impairment (creatinine clearance 15–29 mL/min) is 150 mg orally twice daily. Contraindicated in patients with end-stage renal disease. Pregnancy and Lactation Contraindicated in pregnant women. Women of childbearing potential should use effective contraceptive measures during treatment and for 6 months after discontinuation of the drug. Lactating women should discontinue breastfeeding. Pediatric and Geriatric Populations The safety and efficacy of olaparib in pediatric patients have not been established; use is not recommended. No dose adjustment is required for patients aged 65 years or older, but enhanced monitoring of adverse reactions is needed.
Store in a sealed container, in a dry and well-ventilated place below 25℃. Short-term exposure to temperature fluctuations between 15–30℃ is acceptable. Avoid direct sunlight and humid environments. Keep out of the reach of children.
评论
添加评论
请登录后发表评论
暂无评论