This product is indicated for the treatment of adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) harboring mesenchymal-epithelial transition factor (MET) exon 14 skipping mutations.
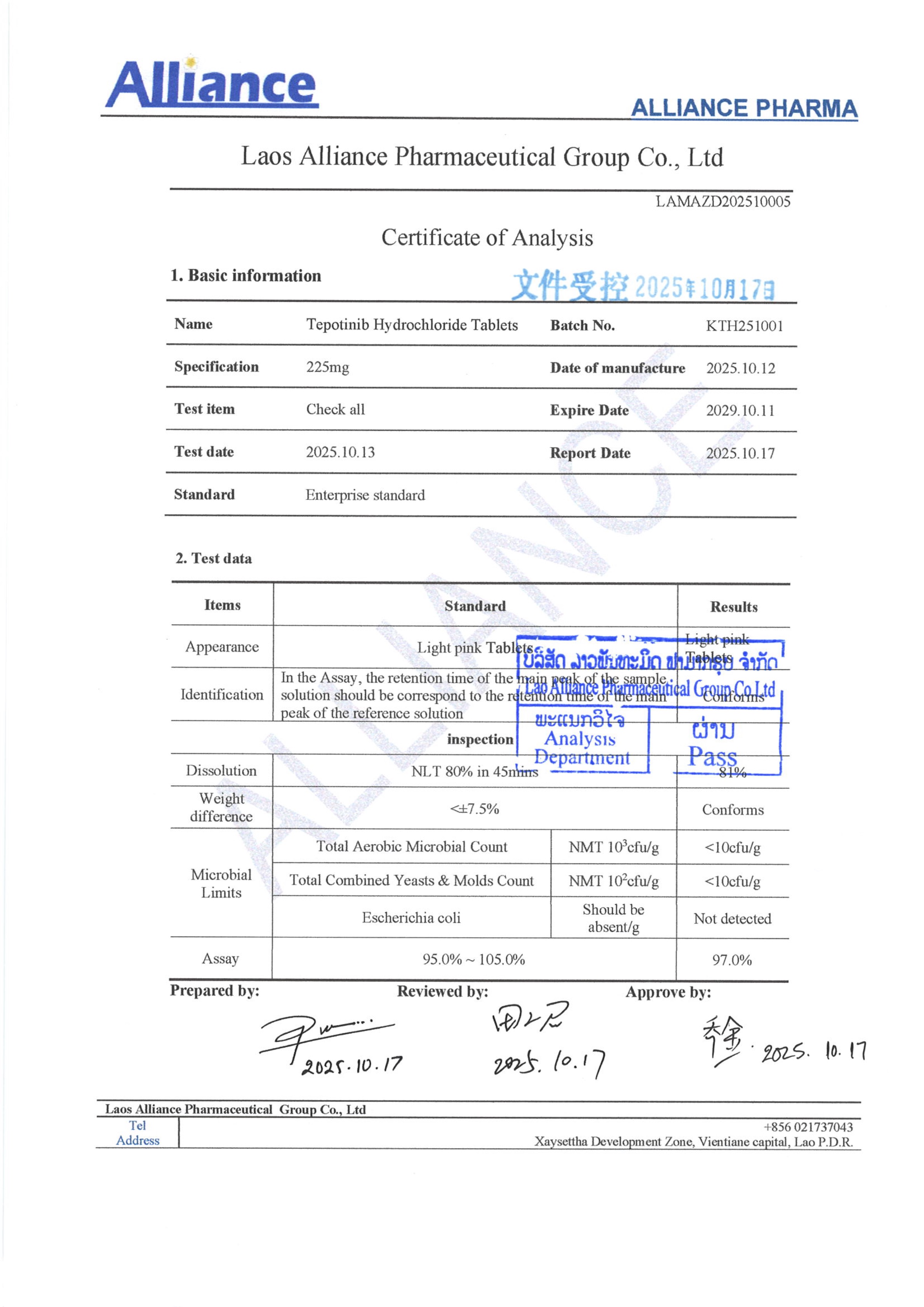
盐酸特泊替尼Tepotinib Hydrochloride 225mg







¥6800.00
售价:¥6800.00
销量:326 件
Patient Selection for METex14 Skipping Mutations Prior to treatment with this product, the presence of MET exon 14 skipping mutations must be confirmed using a fully validated detection method (liquid biopsy or tumor tissue biopsy). Testing plasma specimens for MET exon 14 skipping mutations is recommended only for patients for whom tumor biopsy is not feasible. If no mutation is detected in the plasma sample, re-evaluate the feasibility of tumor tissue testing via biopsy. Recommended Dose The recommended dose is 450 mg (2 tablets) orally once daily, administered with food, until disease progression or unacceptable toxicity occurs. Instruct patients to take this product at approximately the same time each day. Swallow the tablets whole; do not chew, crush or split the tablets. For patients who have difficulty swallowing solids, the tablets may be dispersed in water. If less than 8 hours remain before the next scheduled dose, patients are advised not to take the missed dose. If vomiting occurs after taking a dose, patients are advised to take the next dose at the scheduled time. Dose Adjustment for Adverse Reactions The recommended reduced dose of tepotinib for adverse reactions is 225 mg orally once daily. Permanently discontinue tepotinib in patients who cannot tolerate 225 mg orally once daily. Recommended dose adjustments for tepotinib based on adverse reactions are shown in Table 1. Table 1: Dose Adjustments for Tepotinib Based on Adverse Reactions Adverse Reaction Severity Dose Adjustment Interstitial Lung Disease (ILD)/Pneumonitis Any Grade Temporarily discontinue this product if ILD is suspected. Permanently discontinue this product if ILD is confirmed. Elevated alanine aminotransferase (ALT) and/or aspartate aminotransferase (AST) (without elevated total bilirubin) Grade 3 Temporarily discontinue this product until ALT/AST return to baseline. Resume treatment at the same dose if recovery occurs within 7 days; otherwise, resume at the reduced dose. Grade 4 Permanently discontinue this product. Elevated ALT and/or AST with elevated total bilirubin (without cholestasis or hemolysis) ALT/AST > 3× ULN and total bilirubin > 2× ULN Permanently discontinue this product. Elevated total bilirubin (without elevated ALT and/or AST) Grade 3 Temporarily discontinue this product until bilirubin returns to baseline. Grade 4 Resume treatment at the reduced dose if recovery occurs within 7 days; otherwise, permanently discontinue this product. Increased lipase or amylase Grade 3 Temporarily discontinue this product until levels return to ≤ Grade 2 or baseline. Resume treatment at the reduced dose if recovery occurs within 14 days; otherwise, permanently discontinue this product. Grade 4 Permanently discontinue this product. Pancreatitis Grade 3/4 Permanently discontinue this product. Other adverse reactions Grade 2 Maintain the current dose. If intolerable, consider temporary discontinuation until recovery, then resume at the reduced dose. Grade 3 Temporarily discontinue this product until recovery, then resume at the reduced dose. Grade 4
Interstitial Lung Disease (ILD)/Pneumonitis: Immediately discontinue tepotinib in patients suspected of having ILD/pneumonitis. Permanently discontinue tepotinib in patients diagnosed with ILD/pneumonitis of any severity. Hepatic Toxicity: Monitor liver function tests. Discontinue, reduce the dose, or permanently discontinue tepotinib based on the severity of toxicity. Pancreatic Toxicity: Monitor amylase and lipase levels. Discontinue, reduce the dose, or permanently discontinue tepotinib based on the severity of toxicity. Embryo-Fetal Toxicity: Tepotinib may cause fetal harm.
The main adverse reactions include peripheral edema, hypoalbuminemia, nausea, increased creatinine, and diarrhea.
60 tablets per bottle.
Store tightly closed below 30℃. Keep out of the reach of children.
评论
添加评论
请登录后发表评论
暂无评论