For adult patients with locally advanced or metastatic ROS1-positive non-small cell lung cancer (NSCLC). For adult and pediatric patients aged 12 years and older with locally advanced or metastatic solid tumors harboring neurotrophic tyrosine receptor kinase (NTRK) gene fusions, who have disease progression after prior treatment or have no satisfactory alternative treatment options.
瑞普替尼Repotrectinib 40mg




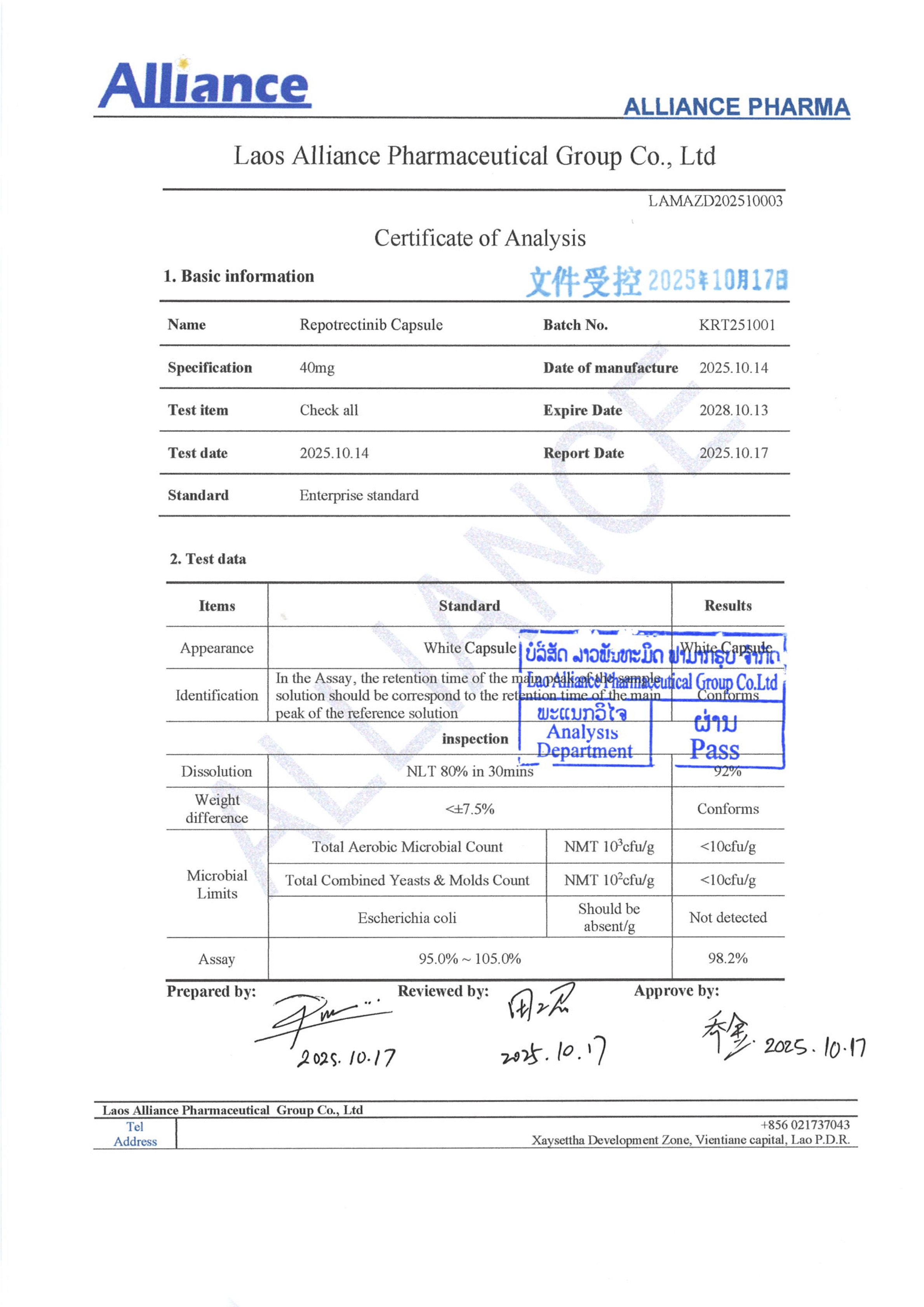

¥1980.00
售价:¥1980.00
销量:52 件
Prior to initiating repotrectinib treatment, discontinue potent and moderate CYP3A inhibitors and allow a washout period equivalent to 3–5 elimination half-lives of the CYP3A inhibitor. Recommended Dose For adult and pediatric patients aged 12 years and older: The initial dose is 160 mg orally once daily for 14 consecutive days. After 14 days, increase the dose to 160 mg orally twice daily, continue treatment until disease progression, unacceptable toxicity, or until surgical resection (which may lead to severe morbidity) is performed. This product may be administered with or without food, at approximately the same time each day. Swallow the capsules whole; do not open, chew, crush, or dissolve the capsules before swallowing. If a dose is missed, or vomiting occurs at any time after taking a dose, skip the missed dose and resume dosing at the next regular scheduled time. Dose Reduction for Adverse Reactions The recommended dose reduction levels for repotrectinib are shown in Table 1. Table 1: Recommended Dose Reduction Levels Initial Dose Dose Reduction Level 1 Dose Reduction Level 2 160 mg once daily 120 mg once daily 80 mg once daily 160 mg twice daily 120 mg twice daily 80 mg twice daily The recommended dose adjustments for specific adverse reactions are shown in Table 2. Table 2: Recommended Dose Adjustments for Adverse Reactions Adverse Reaction Severity Level Dose Adjustment Central Nervous System (CNS) Adverse Reactions Intolerable or Grade 2 Withhold repotrectinib until resolution to Grade ≤1 or baseline. Resume at the same or reduced dose based on clinical need. Grade 3 Withhold repotrectinib until resolution to Grade ≤1 or baseline. Resume at a reduced dose. Grade 4 Permanently discontinue repotrectinib. Interstitial Lung Disease (ILD)/Pneumonitis Any Grade Withhold repotrectinib if ILD/pneumonitis is suspected. Permanently discontinue if ILD/pneumonitis is confirmed. Hepatic Toxicity Grade 3 Withhold repotrectinib until resolution to Grade ≤1 or baseline. Resume at the same dose if improvement occurs within 4 weeks. If Grade 3 event recurs and resolves within 4 weeks, resume at a reduced dose. Grade 4 Withhold repotrectinib until resolution to Grade ≤1 or baseline. Resume at a reduced dose. Permanently discontinue if no improvement within 4 weeks or if Grade 4 event recurs. ALT/AST > 3× Upper Limit of Normal (ULN) with total bilirubin > 1.5× ULN (without cholestasis or hemolysis) - Permanently discontinue repotrectinib. Creatine Phosphokinase (CPK) Elevation CPK > 5× ULN Withhold repotrectinib until resolution to baseline or ≤2.5× ULN; resume at the same dose. CPK > 10× ULN or recurrent CPK > 5× ULN Withhold repotrectinib until resolution to baseline or ≤2.5× ULN; resume at a reduced dose. Hyperuricemia Grade 3/4 Withhold repotrectinib until improvement of signs/symptoms; resume at the same or reduced dose. Other Clinically Relevant Adverse Reactions Intolerable or Grade 2/3/4
Bone Fractures: Promptly evaluate patients who present with signs or symptoms of fractures (e.g., pain, altered mobility, deformity, etc.). Embryo-Fetal Toxicity: May cause fetal harm. Inform females of reproductive potential about the potential risk to the fetus and advise them to use effective non-hormonal contraceptive methods.
The most common adverse reactions (incidence ≥ 20%) are dizziness, dysgeusia, peripheral neuropathy, constipation, dyspnea, fatigue, ataxia, cognitive impairment, myasthenia, and nausea.
60 capsules per bottle.
Store at room temperature, below 30℃. Keep out of the reach of children.
评论
添加评论
请登录后发表评论
暂无评论